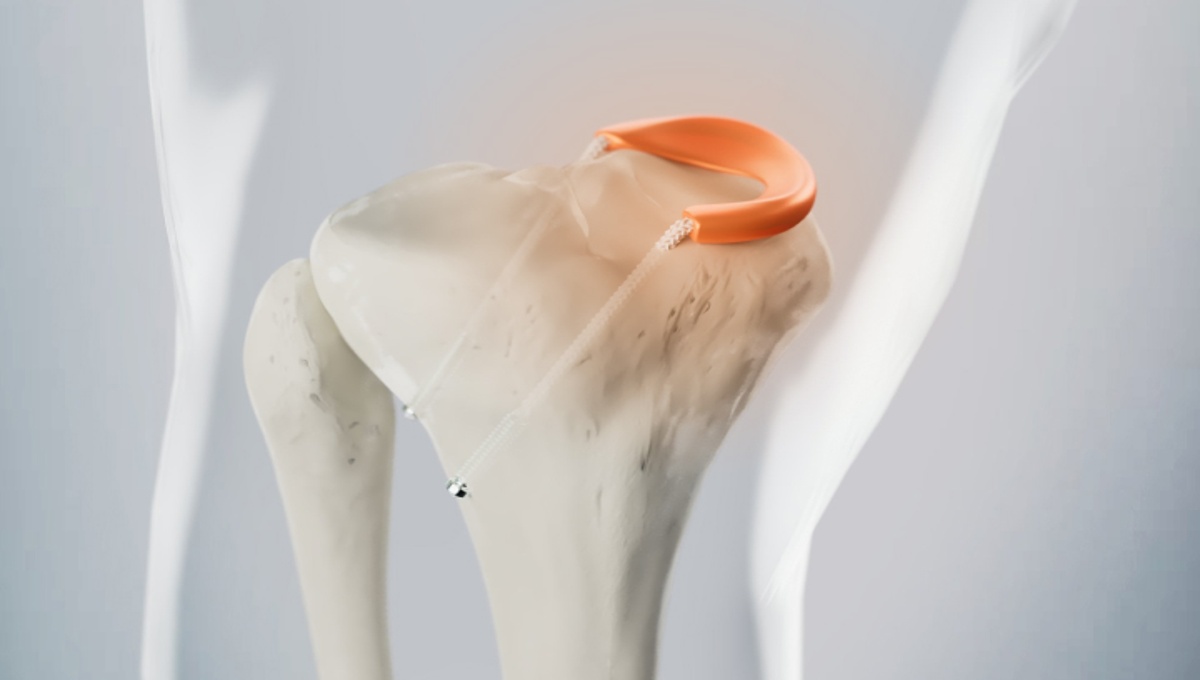
FDA grants breakthrough designation to Orthonika’s synthetic total meniscus implant
Med-Tech Insights reports that Orthonika, a MedTech spin-out from Imperial College London, has received Breakthrough Device Designation from the US FDA for its novel Total Meniscus Replacement (TMR) implant. The technology is designed to replicate the biomechanics of the natural meniscus and restore knee joint function through a minimally invasive arthroscopic procedure.
Meniscal injuries are among the most common orthopaedic procedures worldwide, and in many irreparable cases the current standard treatment involves removing the damaged meniscus. This can lead to long term complications such as reduced mobility and the progression of osteoarthritis. Innovations like Orthonika’s synthetic implant aim to address the gap between meniscus removal and full knee replacement, potentially helping to preserve joint health and delay more invasive surgery.
🔗 Read the full article on Med-Tech Insights: FDA grants breakthrough designation to Orthonika’s synthetic total meniscus implant
Credit for images and content: Med-Tech Insights
____________________________________________________________________________________________________________________________________________________________________________________________________
Cure Talent is a recruitment agency dedicated to the medical technology industry. Whether you are looking for a new role in the medical device sector or you’re an innovative medical device, digital health, or biotech company looking for staff, get in touch with the team today and explore how Cure Talent can help!
