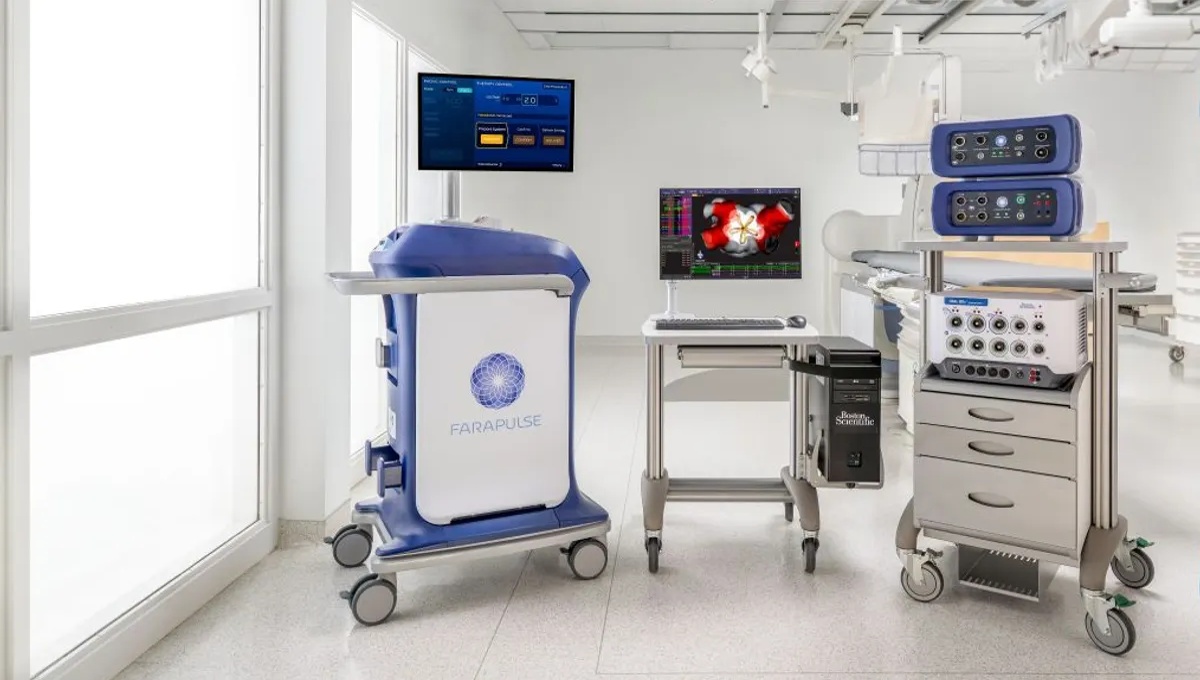
Boston Scientific secures European label expansion for Farapulse system
MedTech Dive reports that Boston Scientific has received CE mark approval in Europe to expand the label for its Farapulse pulsed field ablation (PFA) system. The updated indication allows the technology to be used in patients with persistent atrial fibrillation, a form of irregular heart rhythm that lasts longer than seven days and can increase the risk of stroke.
The approval is supported by data from the ADVANTAGE AF clinical trial, which demonstrated the safety and effectiveness of the system in this patient group. Pulsed field ablation is gaining significant traction in electrophysiology as it offers a more targeted, non thermal approach to cardiac ablation, potentially reducing damage to surrounding tissue compared with traditional techniques.
🔗 Read the full article on MedTech Dive: Boston Scientific gets Farapulse CE mark label expansion in Europe
Credit for images and content: MedTech Dive
____________________________________________________________________________________________________________________________________________________________________________________________________
Cure Talent is a recruitment agency dedicated to the medical technology industry. Whether you are looking for a new role in the medical device sector or you’re an innovative medical device, digital health, or biotech company looking for staff, get in touch with the team today and explore how Cure Talent can help!
